We finally got our neoFroxx periodic table!
Now we can present you our neoFroxx periodic table! You may ask, why are we so excited?
Then you’re probably one of those people who don’t have very exciting memories of the periodic table – lots of tiles on a big board on the wall in the chemistry room and no idea what it all actually means?
We have summarized the most exciting background information for you and presented it graphically. If you want to learn more, then visit us on our social media, where you’ll always find something new and interesting to discover – we look forward to seeing you!
And how do you get a periodic table in neoFroxx design? Simply download it or contact us! We would also be happy to send you a copy in poster size (A2 or A3).

Calcium
The element is important for nutrition and is involved in many biochemical reactions, including the building of skeletal systems, cell signaling and the moderation of muscle activity. It is the most abundant metal in the human body, found mainly in bones and teeth. If you could extract all the calcium in an average adult person, you would have about 1 kg of the metal.

Barium
Always fascinating are the dazzling colors with which the fireworks rockets paint the most amazing shapes on the night sky. But what actually makes New Year’s Eve fireworks so colorful? Barium salts are used, for example, to create green colors in fireworks and to stabilize firework mixtures.

Oxygen
Every oxidation is also a combustion, even respiration. In combustion propulsion, oxygen as an oxidizer is at least as important as the actual fuel. It can be said that most rockets actually owe their propulsion not to the burned fuel, but to the oxygen that is not present in airless space. A jet plane travels a much longer distance on the same amount of fuel than a rocket, which must supply itself with the oxygen it needs.
Phosphorus
Phosphorus is life. What sounds so dramatic, however, is nothing more than a simple fact. Because this element is an essential building block for plants, animals and humans. It already plays an elementary role in our DNA. Together with a pentose, the phosphate groups form the backbone of our genetic material, without which the various bases could not link up.

Carbon
Carbon is one of the most interesting and important elements ever. It even has its own sub-chapter of chemistry dedicated to it – organic chemistry. This deals exclusively with the long carbon chains that this element can exert. We also owe diamonds to carbon. Here, a cirstalline structure is exerted in which each carbon atom is covalently bonded equally with four neighboring atoms. This is why diamonds are so stable.

Einsteinium
Einsteinium was discovered in 1952 after the test of the first American hydrogen bomb and named in honor of Albert Einstein, who personally had nothing to do with the discovery or research on Einsteinium.
What seems to be world-famous, however, is the tongue of our Professor. It was captured on a paparazzi photo about 70 years ago. Today you can still find it a million times on posters, mugs and now also illustrated in our periodic table.

Mercury
Have you ever heard of the hatter’s syndrome? This is a mercury poisoning that often occurred in the 18th century. At that time, it was considered harmless. Today we are wiser.
The whole thing manifested itself as inflammation, nerve damage and restlessness. The organs were attacked from the inside via the blood and the intelligence of the affected persons decreased. The brain damage with personality changes caused by chronic mercury poisoning became known as “mad hatter syndrome” and was immortalized by Lewis Carrol in “Alice in Wonderland”. We join this and give our Hg a hat!


Rutherfordium
This radioactive element, artificially produced in 1964, is dedicated to the experimental physicist and Nobel Prize winner Ernest Rutherford. In addition to his important findings on radioactive decay and half-lives, Rutherford also developed the atomic model, which is still relevant today, in which the electrons of an atom whirl around the positively charged atomic nucleus.
Rhenium
About 20% of the rhenium produced is used for catalysts for unleaded gasoline production. A catalyst made of pure platinum would quickly be oxidized by carbon deposits. The addition of Rhenium avoids this problem and allows conversion at lower temperatures and pressures.

Cobalt
It’s like magic! Materials made of cobalt can be lifted without touching them because of their ferromagnetic properties. What is even more magical, however, is that in the Middle Ages cobalt ores were believed to be bewitched. They were often mistaken for silver and copper ores, but if you tried to melt them down, you would notice a foul-smelling odor. Therefore, goblins were believed to have eaten the valuable ores and excreted goblin ores instead. Over the years, this also gave rise to the name cobalt.

Helium
Helium is the lightest of all elements after hydrogen. Many know it as the content of balloons at fairs. If you fill something with helium, it takes off because it is lighter than air. Sound also spreads much faster in helium than in air. That’s why our voice sounds much higher when we breathe helium. Higher velocity means higher frequency means higher tones. But beware. If you breathe in helium, you expose your body to a lack of oxygen and risk serious health consequences in addition to unconsciousness!
Another fascination of helium is its extremely low boiling point. It is the only element that remains gaseous even at absolute zero under normal pressure!
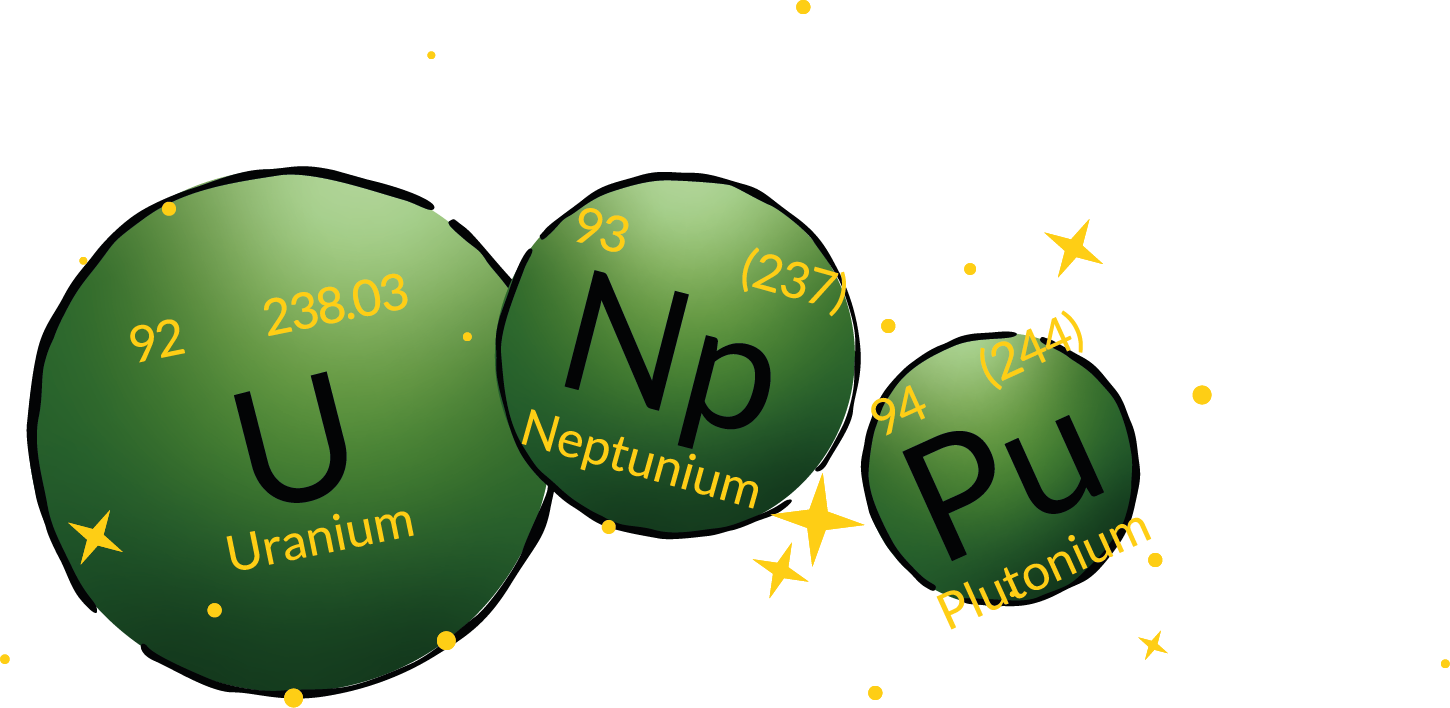
Uranium, neptunium and plutonium
These three elements are named after (dwarf) planets of our solar system.
They follow each other in the periodic as well as in our solar system. All three are toxic, radioactive heavy metals. Uranium is certainly familiar to many from nuclear power plants. There, nuclear fission of uranium takes place and the energy released is converted into electricity. By fissioning 1 kg of uranium, 24,000,000 kWh can be obtained. For comparison: this energy is sufficient to keep a heart beating for 1.8 million years.

